 
(Device Master Record) quality assurance procedures and specifications including control plans, inspection equipment and procedures, acceptance criteria, etc. (Device Master Record) production process specifications including equipment, production methods and procedures, operator (work) instructions, production environment specifications, etc.  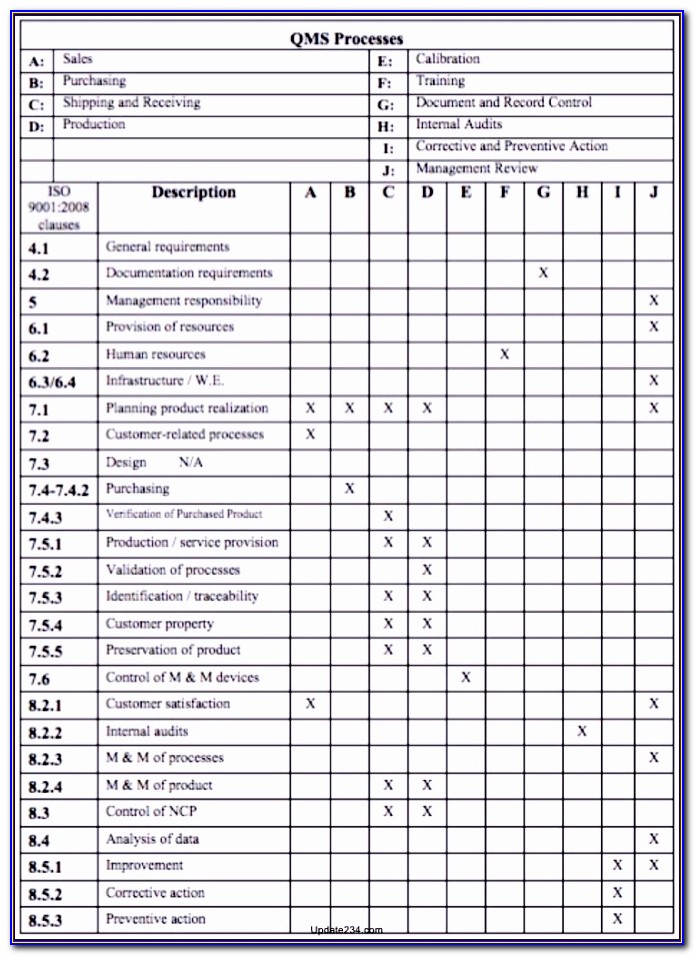 
INTERNAL AUDIT CHECKLIST Issued by: QA ISO13485 Requirements 4 Quality Management System 4.1 General requirements Date: What to look for and how 4.1 Is the quality management system documented, implemented and maintained in accordance with requirements of ISO 13485? Are processes needed for the quality management system identified and established (process map)? Is the sequence and interaction between these processes determined (process map)? Are criteria and methods for the operation and control of quality system processes established (operational procedures)? Are required resources available? Are quality system processes monitored and measured (internal audit, customer feedback, manufacturing process performance, etc.)? 4.1 Are outsourced processes adequately controlled? How are outsourced processes controlled? Are outputs of outsourced processes verified? Are subcontractors and suppliers required to operate and maintain quality management systems (ISO 9001, for example)? 4.2 4.2.1 4.2.1 Documentation requirements General Are the following types of documents established, maintained and controlled: quality policy and quality objectives quality manual operational procedures device specifications including drawings, composition, formulation, components, software etc. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
